Patient Highlights
- Post-transplant clonoSEQ Tracking (MRD) Testing revealed measurable residual disease (MRD); flow cytometry results did not detect measurable residual disease
- MRD continued to decline until patient became MRD-negative
- Patient continues to be monitored for MRD with peripheral blood samples
Patient History
- Patient received pediatric induction regimen followed by consolidation therapy
- Pre-transplant flow cytometry MRD was negative and clonoSEQ Tracking (MRD) Test was MRD-positive. Patient received allogenic stem cell transplant
- Immunosuppression therapy was discontinued post-transplant
- Patient continues to be monitored by clonoSEQ Tracking (MRD) Tests with peripheral blood samples
Physician's Perspective
“This patient was told that he was MRD-negative in the bone marrow by flow cytometry when he was referred to me for transplant. My standard of care is to take a peripheral blood clonoSEQ Tracking (MRD) test prior to transplant. This test showed that the patient was MRD-positive. My experience is that patients who are MRD-negative prior to transplant have better outcomes post-transplant.” *
*Clinician has received compensation to participate in advisory meetings sponsored by Adaptive. Clinician’s research has also been supported, in part, via product grants.
Use of the clonoSEQ Assay
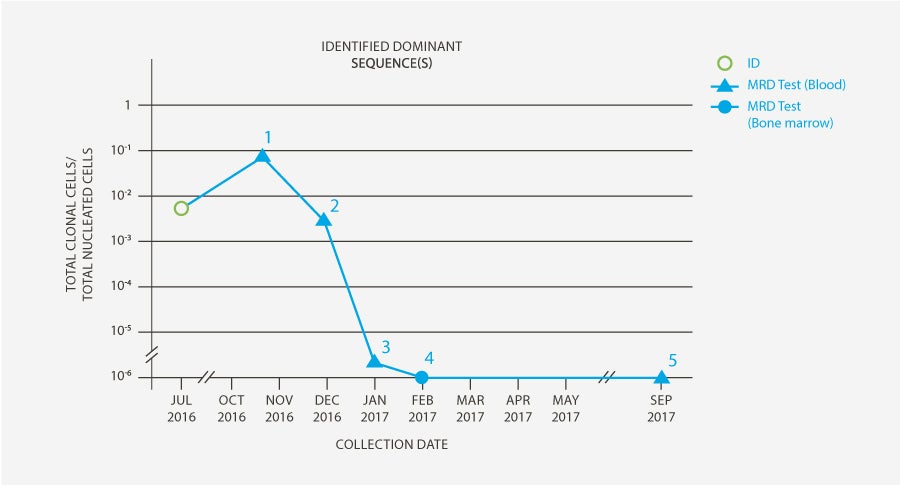
-

Prior to transplant, the patient was MRD-negative in the bone marrow by flow cytometry. The patient was MRD-positive in the peripheral blood by clonoSEQ. The patient then received an ASCT in November 2016.
-

Day 28 post-transplant, the patient continued to have a high level of MRD present in the peripheral blood. Due to this, immunosuppression was discontinued.
-

Day 60 post-transplant, the clonoSEQ (MRD) Tracking Test detected only 1 malignant cell in 1,000,000 total nucleated cells.
-

To confirm response in the bone marrow, MRD was assessed by clonoSEQ Day 90 post-transplant. The result was MRD-negative.
-

MRD will continue to be monitored by the clonoSEQ Tracking (MRD) Test in the peripheral blood. About 11 months posttransplant, the patient continues to be MRD-negative in the peripheral blood.
Blood-based MRD testing for patients with acute lymphoblastic leukemia is available as a CLIA-regulated laboratory developed test (LDT) service provided by Adaptive Biotechnologies. This use of clonoSEQ has not been approved or cleared by the FDA.
*This case study was based off results generated from an earlier version of the clonoSEQ Assay.
Intended Use:
The clonoSEQ Assay is an in vitro diagnostic that uses multiplex polymerase chain reaction (PCR) and next-generation sequencing (NGS) to identify and quantify rearranged IgH (VDJ), IgH (DJ), IgK and IgL receptor gene sequences, as well as translocated BCL1/IgH (J) and BCL2/IgH (J) sequences in DNA extracted from bone marrow from patients with B-cell acute lymphoblastic leukemia (ALL) or multiple myeloma (MM), and blood or bone marrow from patients with chronic lymphocytic leukemia (CLL).
The clonoSEQ Assay measures minimal residual disease (MRD) to monitor changes in burden of disease during and after treatment. The test is indicated for use by qualified healthcare professionals in accordance with professional guidelines for clinical decision-making and in conjunction with other clinicopathological features.
The clonoSEQ Assay is a single-site assay performed at Adaptive Biotechnologies Corporation in Seattle, Washington.
Special Conditions for Use:
- For in vitro diagnostic use.
- For prescription use only (Rx only).
Limitations:
ALL, MM, and CLL:
MRD values obtained with different assay methods may not be interchangeable due to differences in assay methods and reagent specificity. The results obtained from this assay should always be used in combination with the clinical examination, patient medical history, and other findings. The clonoSEQ Assay is for use with specimens collected in EDTA tubes. Results may vary according to sample time within the course of disease or by sampling site location. The assay may overestimate MRD frequencies near the limit of detection (LoD). The MRD frequency LoD varies based on the amount of gDNA that is tested and using lower gDNA input may prevent MRD detection at low frequencies. Sample processing and cell enrichment strategies may affect the measured MRD frequency. The volume and cellularity of sampled input material may affect the ability to detect low levels of disease. False positive or false negative results may occur for reasons including, but not limited to: contamination; technical and/or biological factors such as the type of rearrangement or the size of the junction region. The assay has been validated with the Illumina NextSeq500 and 550.
For CLL:
MRD is based on measurements of tumor cells detected in peripheral blood and/or bone marrow. However, patients may have significant residual disease in unassessed compartments and U-MRD in one compartment cannot fully rule out the presence of disease in the other compartment, for example, U-MRD in blood may not be the same in bone marrow. Therefore assessment of MRD in CLL should employ a multimodal approach including clinical examination, patient medical history, and other findings. Outcome for patients with MRD detectable in bone marrow but not in peripheral blood (PB-/BM+) may differ according to type of therapy. This assay is capable of monitoring specific tumor clonotypes. The association between MRD assessments and patient clinical status for the purpose of monitoring changes in disease (e.g., relapse, remission, stable disease) has not been demonstrated. The value of MRD in CLL for previously untreated or “watch and wait” patients is not established. CLL is a heterogeneous disease. MRD values and expectations for outcome may not be generalizable across treatments. Changes in MRD should be interpreted with caution when used to evaluate disease burden in therapies that have not been validated. Regardless of MRD status, cytogenetics play an independent role in patient risk status and its impact on PFS/OS.
For important information about the FDA-cleared uses of clonoSEQ including test limitations, please visit clonoSEQ.com/technical-summary.
 Back
Back